Unité — Interactions Arbres/Micro-organismes (IAM)
Chloroplast FBPase and SBPase are thioredoxin-linked enzymes with similar architecture but different evolutionary histories
Desirée Gütle, Thomas Roret, Stefanie Mueller, Jérémy Couturier, Stéphane Lemaire, Arnaud Hecker, Tiphaine Dhalleine, Bob Buchanan, Ralf Reski, Oliver Einsle, Jean-Pierre Jacquot. June 2016. Proceedings of the National Academy of Sciences (USA) – PNAS
___________________________________________________________________
Resumé (en anglais)
Significance
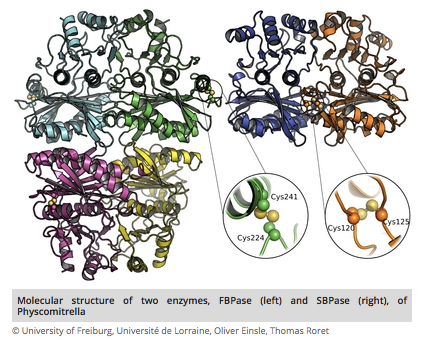
We demonstrate that, although the two phosphatases of the Calvin–Benson cycle of photosynthesis [sedoheptulose-1,7-bisphosphatase (SBPase) and fructose-1,6-bisphosphatase (FBPase)] share extensive structural homology, their redox-regulatory disulfides are incorporated in strikingly different positions, in agreement with an independent evolutionary origin of each enzyme. This article compares in detail the structures of the enzymes together with their regulatory and catalytic properties as well as their phylogenies. Significantly, the substrate binding site of SBPase is larger than that of FBPase, thus allowing it to accommodate both seven- and six-carbon sugar phosphate substrates, whereas FBPase is active only with the latter. The data suggest that SBPase is of archaeal origin, whereas FBPase is descended from bacteria.
Abstract
The Calvin–Benson cycle of carbon dioxide fixation in chloroplasts is controlled by light-dependent redox reactions that target specific enzymes. Of the regulatory members of the cycle, our knowledge of sedoheptulose-1,7-bisphosphatase (SBPase) is particularly scanty, despite growing evidence for its importance and link to plant productivity. To help fill this gap, we have purified, crystallized, and characterized the recombinant form of the enzyme together with the better studied fructose-1,6-bisphosphatase (FBPase), in both cases from the moss Physcomitrella patens (Pp). Overall, the moss enzymes resembled their counterparts from seed plants, including oligomeric organization—PpSBPase is a dimer, and PpFBPase is a tetramer. The two phosphatases showed striking structural homology to each other, differing primarily in their solvent-exposed surface areas in a manner accounting for their specificity for seven-carbon (sedoheptulose) and six-carbon (fructose) sugar bisphosphate substrates. The two enzymes had a similar redox potential for their regulatory redox-active disulfides (−310 mV for PpSBPase vs. −290 mV for PpFBPase), requirement for Mg2+ and thioredoxin (TRX) specificity (TRX f > TRX m). Previously known to differ in the position and sequence of their regulatory cysteines, the enzymes unexpectedly showed unique evolutionary histories. The FBPase gene originated in bacteria in conjunction with the endosymbiotic event giving rise to mitochondria, whereas SBPase arose from an archaeal gene resident in the eukaryotic host. These findings raise the question of how enzymes with such different evolutionary origins achieved structural similarity and adapted to control by the same light-dependent photosynthetic mechanism—namely ferredoxin, ferredoxin-thioredoxin reductase, and thioredoxin.